NovaCina, a trusted CDMO, provides premium sterile liquid pharmaceutical contract manufacturing services. With expertise spanning early-stage drug development, full-scale manufacturing, and advanced analytical studies, we empower you to deliver market-leading products of unparalleled quality.
We specialise in sterile injectables
High Potency
Dramatically shift the paradigm in cytotoxic and innovative drug treatments using our multi-format dosing capabilities in ready-to-use plastic vials. Approved and licensed to manufacture products for 72 countries.
Blow Fill Seal
Safest unit dose every time, matched with unrivaled, cost-effective, high-speed manufacturing techniques. Capabilities to service all batch sizes of any scale, from larger to small.
Multi format vials
Extensive selection of vials available glass or plastic in a range of sizes for both oxygen and non-oxygen sensitive products
Services
NovaCina is a highly experienced and reliable partner focused on speed and extremely responsive customer service. We offer a range of technical and analytical services for every stage of your product development needs.
Our Facility

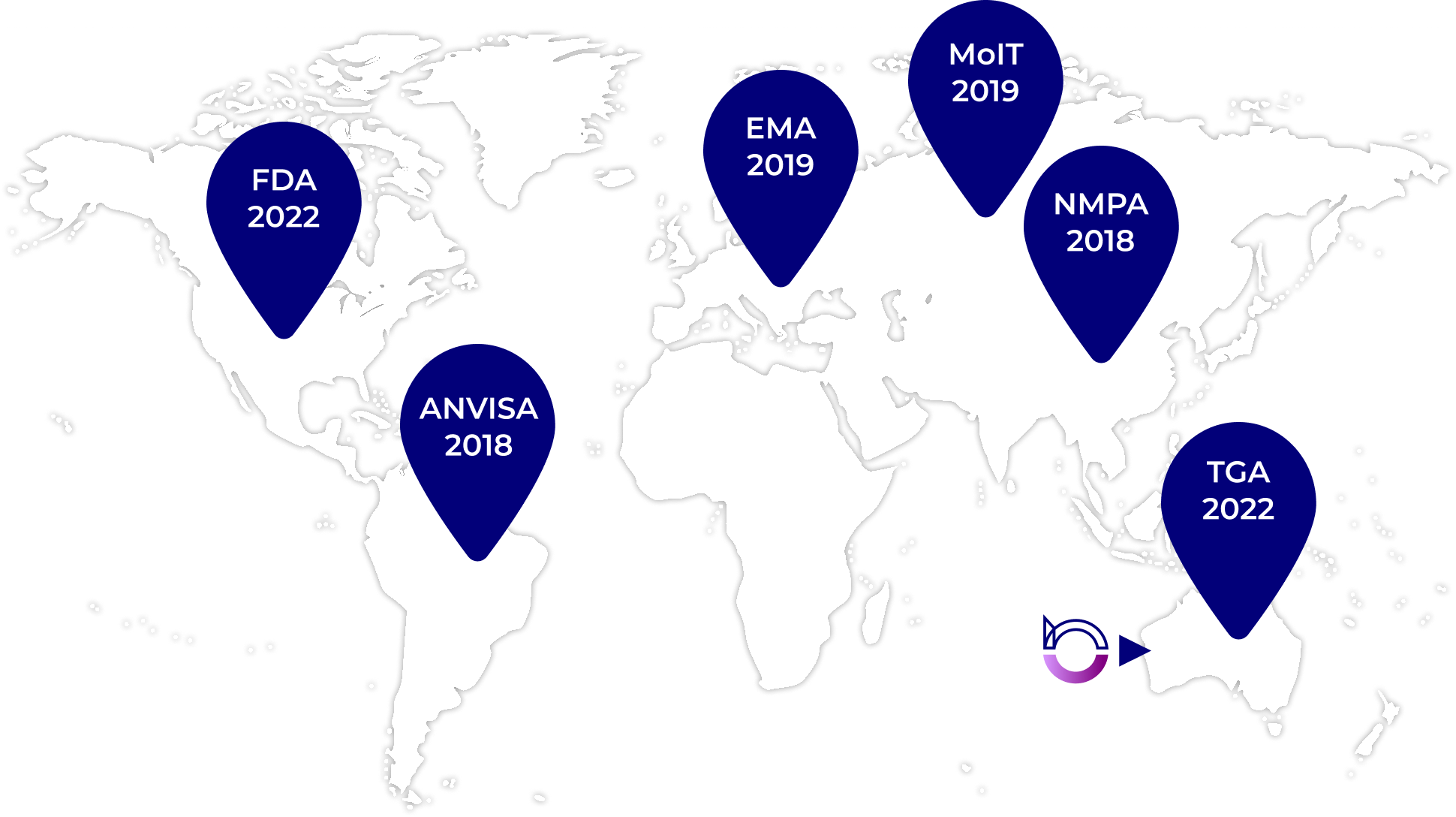
We combine highly automated manufacturing with process excellence to deliver high-quality results to our customers with a high degree of reliability. Over its three decade history, our site has an excellent track-record of supply reliability and delivery consistency. We have an industry leading track record in all areas of regulatory compliance as demonstrated by the multitude of regulatory inspections performed by global health authorities. Throughout its history, the site has never been issued a warning letter, including over the past five years when inspections have been undertaken by the FDA (United States), TGA (Australia), and ANVISA (Brazil).
Service Offerings
NovaCina is a reliable and flexible partner to help your product scale the development ladder from discovery stage to final product manufacturing. We offer a full suite of technical support, analytical testing, and microbiology testing services that can be custom tailored for your specific needs. NovaCina’s highly trained and long-tenured technical staff has decades of experience formulating sterile and non-sterile liquid products and commercial presentations for international export. Our team prides itself on its speed and highly responsive customer service, ensuring that your products make it to the market ahead of schedule.

Regulatory & Compliance
Our facility has an excellent track record when it comes to regulatory compliance and has recently passed inspections by multiple international regulatory bodies.
Founders
NovaCina is proudly owned by the Bridgewest Group. The Bridgewest Group is an investment company with a passion for innovation in technology and life sciences. For over 20 years, we have developed businesses with innovative solutions that improve the lives of patients and advance the field of medicine. With a team of highly skilled scientists and researchers, the Bridgewest Group is committed to conducting cutting-edge research and using the latest technologies to bring new and effective drugs to market.
Bridgewest is committed to building Australia’s pharmaceutical and biological manufacturing industry, providing high quality and high paying employment opportunities to Australians, and helping Australia build a more resilient medical supply chain. Bridgewest is at the forefront in helping transition Australia from an R&D hub to an end-to-end pharmaceutical powerhouse, complete with world-class manufacturing capabilities. NovaCina is the second major contract manufacturing and development (CDMO) investment Bridgewest has made in Australia. Bridgewest is also a proud investor in BioCina, based in Adelaide, which is a leader in microbial drug development and the only biologics focused GMP facility in Australia to be approved by the United States’ FDA.











